Lung cancer treatment receives FDA approval following UCLA trials
By Allison Ong
Nov. 3, 2016 11:31 p.m.
Four years ago, lung cancer patients began traveling to Westwood from as far away as Hawaii and Tennessee to test the latest potential treatment for their disease.
Oncology researchers and doctors at the UCLA Jonsson Comprehensive Cancer Center were conducting the phase one clinical trial of a drug called pembrolizumab, which had demonstrated success in treating the skin cancer melanoma. Last month, the drug – marketed as Keytruda – received first-line approval from the Food and Drug Administration for the treatment of non-small cell lung cancer, or NSCLC.
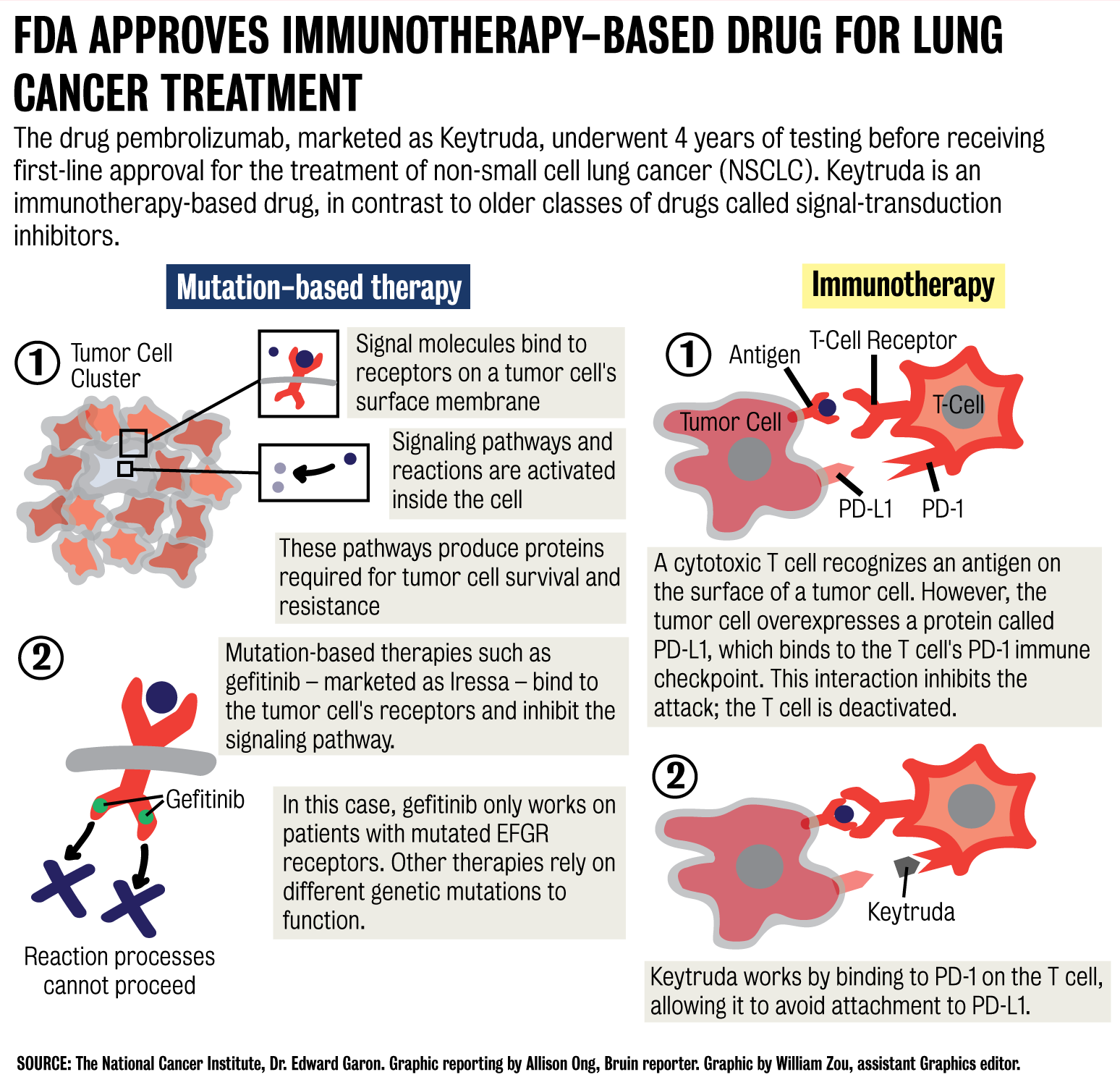
The FDA’s first-line approval will allow people with NSCLC to choose pembrolizumab as their initial treatment instead of standard chemotherapy, said Edward Garon, director of thoracic oncology at JCCC. Pembrolizumab combats tumors by harnessing the power of cytotoxic T cells from the patient’s immune system. The drug, classified as a PD-1 inhibitor, is an alternative to chemotherapy and mutation-based therapies, which fight tumor cells by blocking their intracellular chemical signaling pathways.
NSCLC constitutes 85 percent of all lung cancer cases in the U.S. The cancer subtype attacks epithelial cells between the lung’s bronchial tubes and alveoli, little air sacs in the lung where gases exchange during respiration. Smoking is the largest risk factor for this type of cancer, with a five-year survival rate of approximately 20 percent.
UCLA researchers first tested pembrolizumab on patients in May 2012. The study grew to include 495 patients at multiple sites.
“From the perspective of drug development, this is a very rapid movement of a drug from first patient to full approval in a large population,” Garon said.
Pembrolizumab essentially inhibits a tumor cell’s defensive mechanism. Healthy cells often express a protein called programmed death ligand 1, or PD-L1, on their external surfaces, Garon said. The interaction between PD-L1 and programmed death 1, or PD-1 – a protein found on immune system T cells – prevents the T cells from destroying healthy cells.
[Related: UCLA researchers develop new immunotherapy for advanced brain cancer]
But diseased tumors can also express high levels of PD-L1 on their surfaces, allowing them to interact with PD-1 and resist immune system destruction, Garon said. Pembrolizumab works by blocking these interactions between PD-L1 and PD-1, enabling T cells to target and kill tumors as they should.
In their first trial, UCLA researchers administered pembrolizumab to all patients under their care. Those with higher levels of PD-L1 expression reacted more favorably to the drug than any other group, Garon said.
“By enhancing for this biomarker (PD-L1), the pembrolizumab did identify a group of patients that did better than those on chemo, and considerably,” he said.
About 80 percent of patients in most of the studies were smokers, said Jonathan Goldman, the director of clinical trials at JCCC and an assistant professor in hematology-oncology. But the pool was diverse; some led physically and mentally active lives while others joined as more of a last-ditch effort to find treatments that worked for them, he said.
“I remember a patient in his late 70s who was really quite discouraged, but was advised by his local doctor to see what options he had (for treatment),” Goldman said. “Very quickly, the patient had a greater than 90 percent shrinkage of his tumor within nine weeks.”
Besides lung and skin cancers, pembrolizumab has demonstrated clinical success in treating other cancer such as head and neck cancer, bladder cancer, colorectal cancer and Hodgkin’s lymphoma, Garon said. He and other researchers want to continue their research on pembrolizumab drug combinations, and develop more treatments for patients with specific genetic mutations.
[Related: UCLA student inspired by Ellen DeGeneres, fights cancer with humor]
JCCC typically runs 100 to 150 active trials for cancer patients, said Peter Bracke, communications director for JCCC. Researchers have played integral roles in developing Herceptin, the first targeted anti-breast cancer drug, and Gleevec, a daily pill for those with leukemia.
“The focus of cancer research at UCLA has always been about taking basic science discoveries to the clinic,” Bracke said. “Over 400 scientists and physicians conduct basic and translational cancer research here.”
Goldman said the opportunity to make dramatic changes in lung cancer treatment was a once-in-a-lifetime experience. But the area still requires a lot of research, resources and intention to move forward.
“Some of these new therapies really bring a lot of hope to patients, which is wonderful,” he said. “But it was also difficult to see them coming in with such hope, and not everyone benefiting. … That’s why we’re working so hard to try and expand the benefits of immunotherapy to other patients.”

